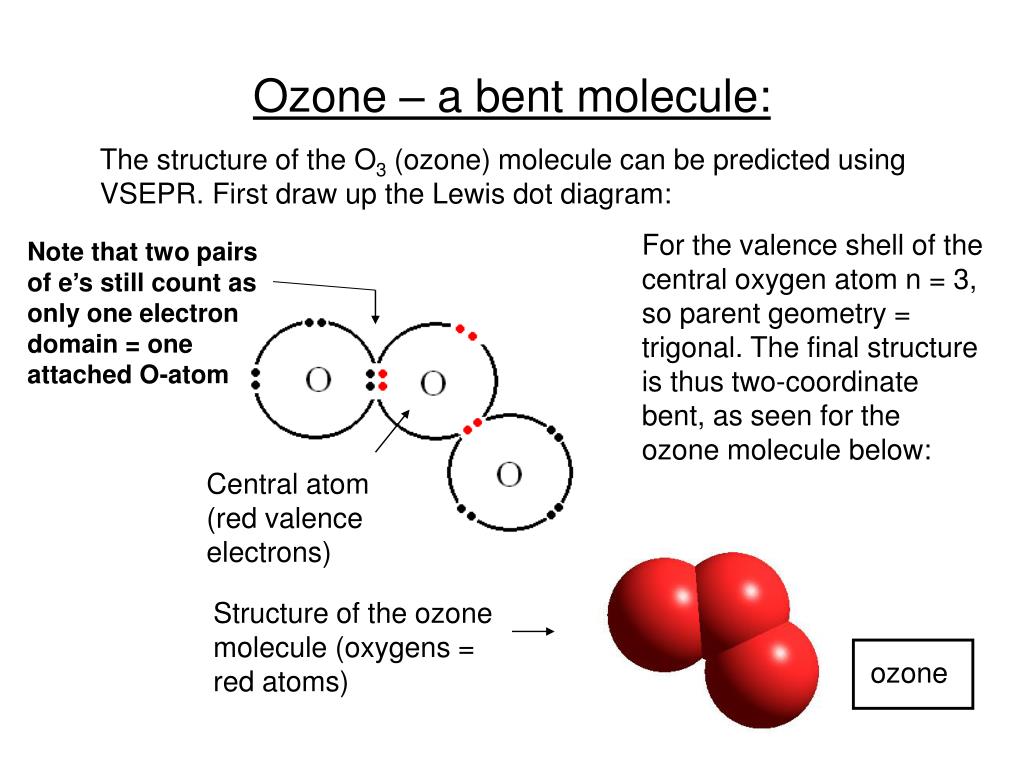
 valence electrons of oxygen in SO2 are 6.The three pairs of bonding electrons are arranged in the plane at an angle of 120-degree.The molecular geometry of SO 2 is a trigonal planner.The total number of SO2 valence electrons is 12. The number of valence electrons in both S and O atoms is six. SO2 Lewis structure would comprise two atoms of oxygen (O) and one sulfur atom. More Links Is BF3 Polar or Nonpolar? CO Lewis Structure & Molecular Geometry CH4 Lewis Structure & Molecular Geometry N2 Lewis Structure| Hybridization & Molecular Geometry BF3 Lewis structure| Molecular geometry, Hybridization Molar Mass of Acetic Acid HCN Lewis Structure & Molecular Geometry Frequently Asked Questions (FAQs) 1. O3 is a polar molecule with a bent trigonal planar shape.The bond angle is 116.8 degrees and there are 18 valence electrons.In the O3 Lewis structure, there is a double bond between the central oxygen atom and one lateral oxygen atom.To summarize everything in this article, the following are some important points: Higher stratospheric ozone concentrations are thus important to ensure that life (including humans) on Earth’s surface is not exposed to hazardous UV-B radiation concentrations. The ozone layer typically absorbs 97-99 percent of incoming UV-B radiation. It covers the whole globe and preserves life by absorbing the sun’s damaging ultraviolet-B (UV-B) rays. The phrase “ozone layer” refers to the high concentration of ozone found in the stratosphere about 15–30 km above the earth’s surface. Ozone is created when heat and sunlight cause chemical reactions between nitrogen oxides (NOX) and volatile organic compounds (VOC), sometimes known as hydrocarbons. Ozone, abbreviated as O3, is a molecule made up of three oxygen atoms. 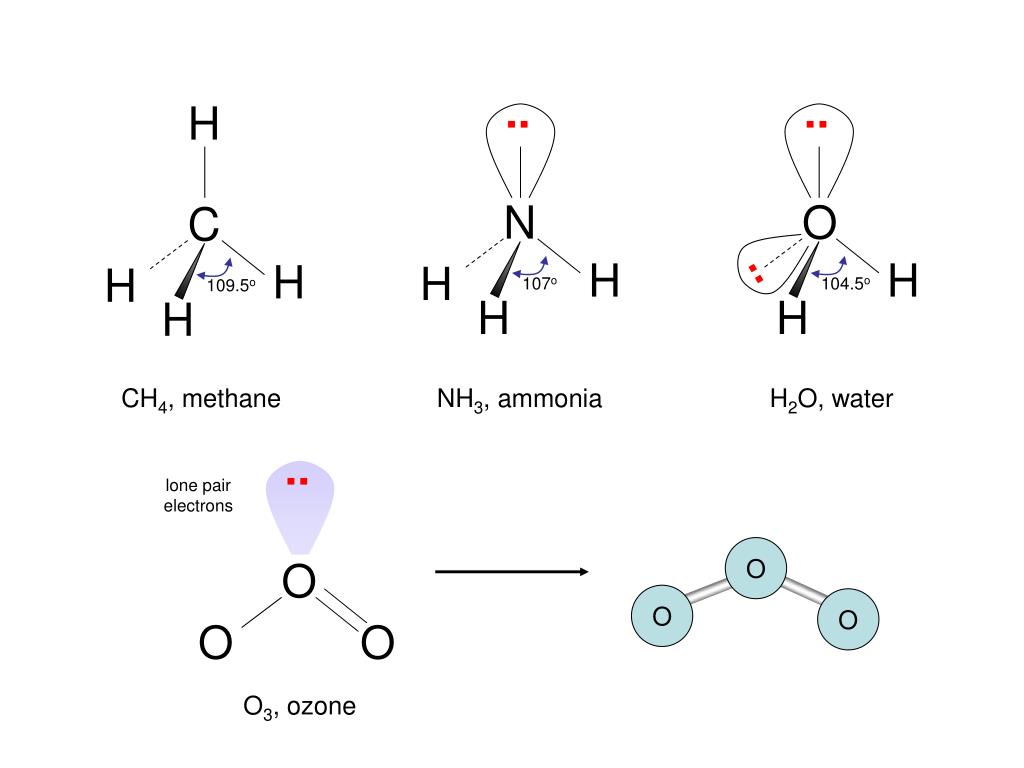
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
